Methanol Structure, Density & Molar Mass - Video & Lesson
4.6 (681) · € 27.00 · Auf Lager
If the density of methanol is 0.793 kg/L what is its volume needed for making 2.5L of its 0.25 molar solution? - Quora

SOLVED: Q.2 A mixture of ethanol (ethyl alcohol) and water contains 60.0% water by mass. (a) Assuming volume additivity of the components, estimate the specific gravity of the mixture at 20°C. What
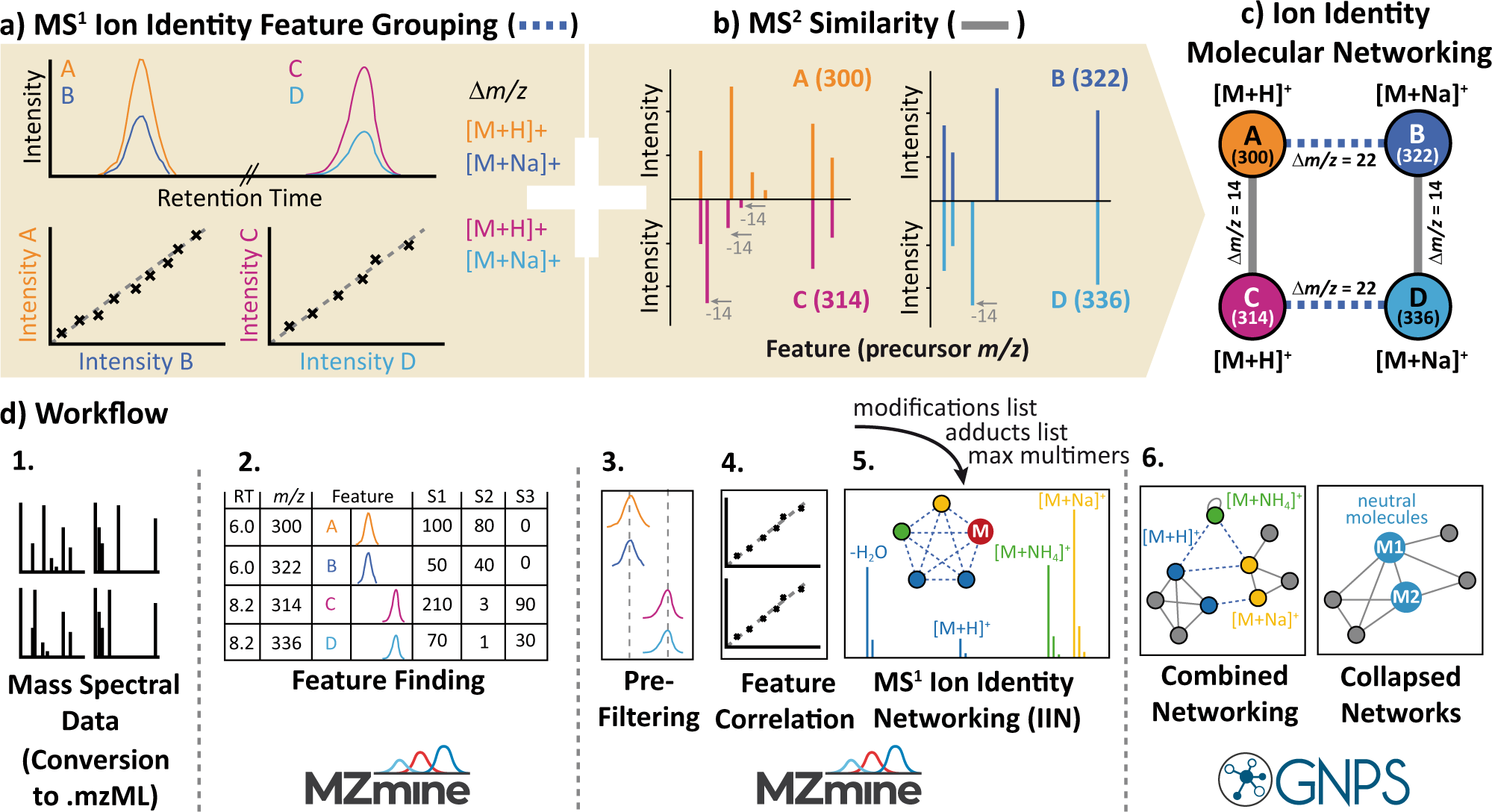
Ion identity molecular networking for mass spectrometry-based metabolomics in the GNPS environment

Important Questions for Class 12 Chemistry Chapter 11 - Alcohols, Phenols and Ethers Class 12 Important Questions
If the density of methanol is 0.793 kg/L what is its volume needed for making 2.5L of its 0.25 molar solution? - Quora

If the density of methanol is 0.793 kg/L.What is its volume needed for making 2.5L of its 0.25M sol?

Acetone, Structure, Uses & Formula - Video & Lesson Transcript

What is the molecular weight of methanol?, by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry.

Molar Heat of Combustion, Definition, Equation & Formula - Video & Lesson Transcript

Methanol, Structure, Density & Molar Mass - Video & Lesson Transcript

Influence of methanol molarity on DMFC polarization (empty symbols) and

Hexane, Formula, Structure & Uses - Video & Lesson Transcript

How to Find Molar Mass of Methanol, What is the Molecular Weight of Methanol CH3OH?