Secretion mechanism of the autotransporter proteins. (A) Structure of
5 (546) · € 26.50 · Auf Lager
Download scientific diagram | Secretion mechanism of the autotransporter proteins. (A) Structure of the polyprotein precursor. (B) Transport of the recombinant passenger. By the use of a typical signal peptide, a precursor protein is transported across the inner membrane. After arrival at the periplasm, the C-terminal part of the precursor folds as a porin-like structure, a so-called  -barrel within the outer membrane, and the passenger is transmitted to the cell surface. SP, signal peptide; IM, inner membrane; PP, periplasm; OM, outer membrane. from publication: The Autodisplay Story, from Discovery to Biotechnical and Biomedical Applications | Among the pathways used by gram-negative bacteria for protein secretion, the autotransporter pathway represents a solution of impressive simplicity. Proteins are transported, independent of their nature as recombinant or native passengers, as long as the coding nucleotide | Biomedical Applications, Escherichia Coli Adhesins and Neisseria gonorrhoeae | ResearchGate, the professional network for scientists.

From self sufficiency to dependence: mechanisms and factors important for autotransporter biogenesis

Type V secretion: mechanism(s) of autotransport through the bacterial outer membrane

Schematic representation of the type I, II, III, and IV protein
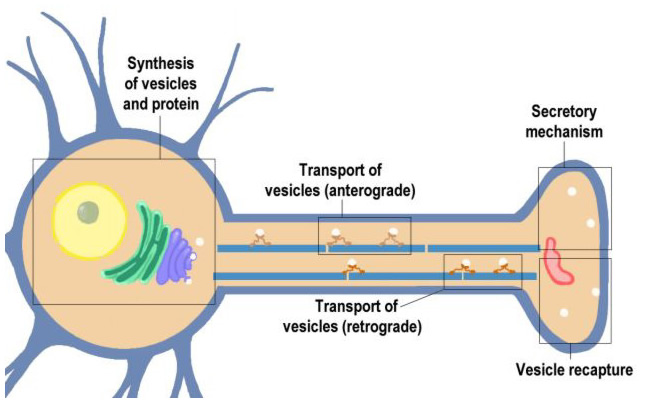
Transport and the Molecular Mechanism of Secretion (Section 1, Chapter 10) Neuroscience Online: An Electronic Textbook for the Neurosciences
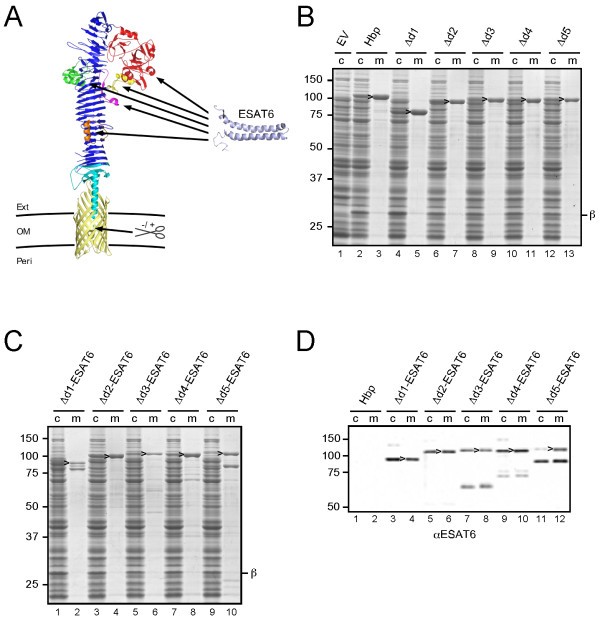
A structurally informed autotransporter platform for efficient heterologous protein secretion and display, Microbial Cell Factories

Alternative mechanisms of autotransporter protein secretion. (A)

Antibiotics, Free Full-Text

The great escape: structure and function of the autotransporter proteins: Trends in Microbiology

Efficient Autotransporter-Mediated Extracellular Secretion of a Heterologous Recombinant Protein by Escherichia coli

Antibiotics, Free Full-Text

Autotransporter proteins, evolution and redefining protein secretion: Trends in Microbiology
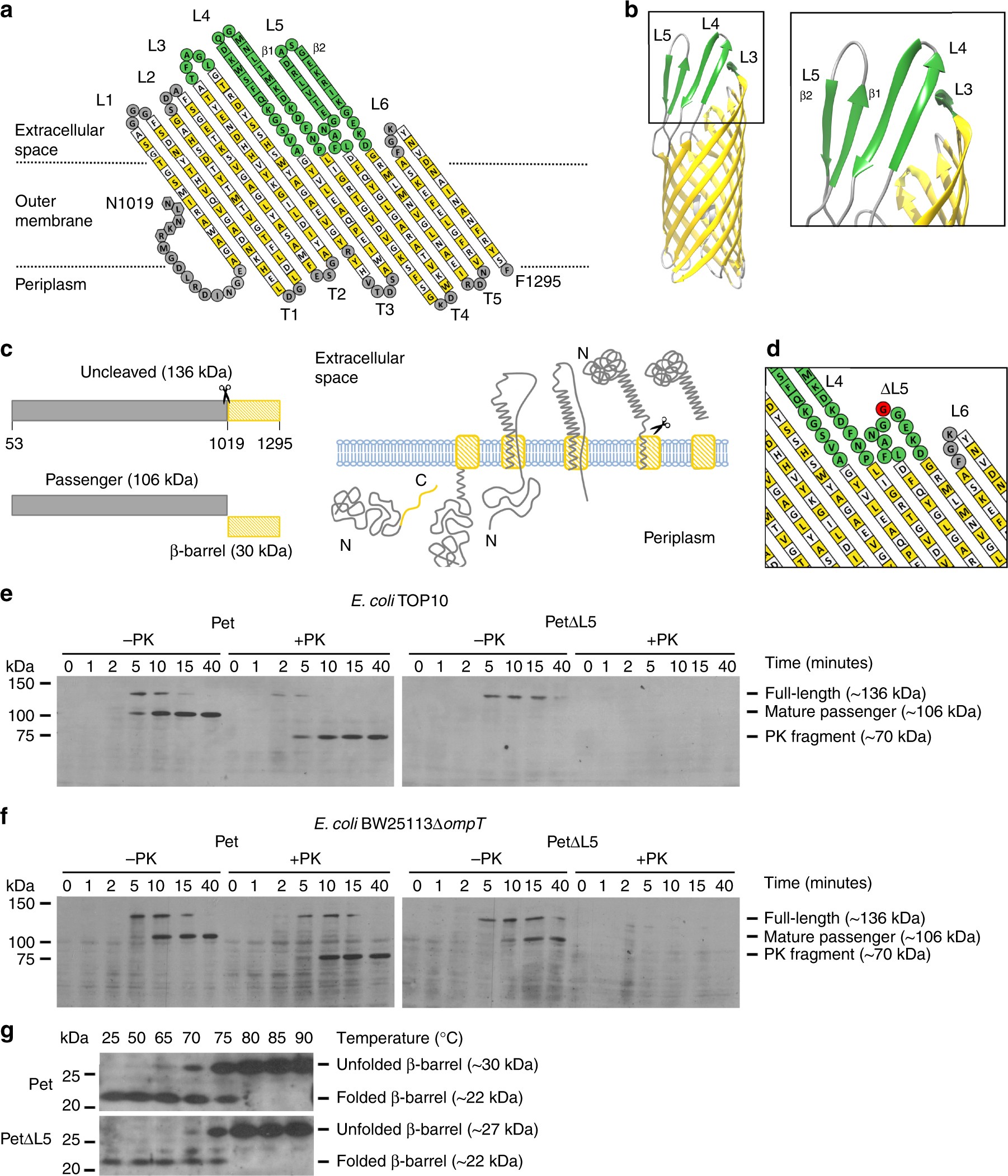
Molecular basis for the folding of β-helical autotransporter passenger domains
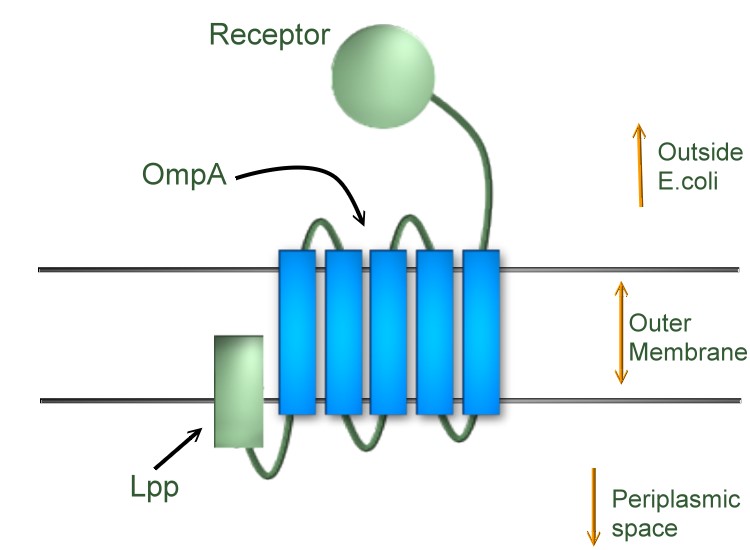
Team:NYMU-Taipei/Project/Receptor/Autotransporter Secretion System












